
Transoral fundoplication for gastroesophageal reflux disease
Transoral incisionless fundoplication has been introduced in clinical practice in the 2007 and mimicks the partial circumferential or anterior fundoplication for treatment of gastro-esophageal reflux disease (1-4). The technique reconfigures the tissue to obtain a full-thickness gastro-esophageal valve from inside the stomach, by serosa-to-serosa plications which include the muscle layers: the new valve is capable to boost the barrier function of the lower esophageal sphincter (LES) with patient’s less discomfort and possibly fewer technique-related complications and side effects, compared to surgery.
TIF may be performed by using the EsophyX™ device (EndoGastric Solutions, Redmond, WA, USA) or the Medigus ultrasonic surgical endostapler (MUSE™, Medigus, Omer, Israel). The endoluminal platform with the greatest global experience so far is the TIF performed by using the EsophyX device. EsophyX constructs an omega-shaped valve 3–5 cm long, in a 250–300 circumferential pattern around the gastro-esophageal junction, by deploying multiple non-absorbable polypropylene fasteners through the two layers (esophagus and stomach) under endoscopic vision of the operator. TIF with this device has proved good, durable, long-term follow-up data from multiple investigators that have used the TIF-2 technique. The device has been recently updated and improved in a new generation instrument: the EsophyX Z.
Medigus ultrasonic surgical endostapler (MUSE™) represents the newest endoluminal fundoplication device approved by FDA. MUSE staples the fundus of the stomach to the esophagus below the diaphragm using multiple sets of metal stitches placed under an ultrasound-guided technique and creates an anterior fundoplication functionally similar to standard surgical Dor-Thal operation. Differently from EsophyX, the new valve is constructed under ultrasonic control. In the case of sliding hiatal hernia, the procedure can be performed only if the hernia can be reduced below the diaphragm.
To date, after 10 years of clinical practice and more than 20,000 procedures performed, TIF may be proposed as an effective and promising therapeutic option in alternative to medical and surgical therapy in patients suffering from reflux esophagitis, hypersensitive esophagus, and Barrett’s esophagus with either competent gastro-esophageal valve or hiatal hernia not exceeding 3 cm in length, who refuse, are intolerant or unresponsive to proton pump inhibitor (PPI) maintenance therapy.
Current issues about TIF refer to: (I) the capability of the procedure to effectively control gastroesophageal reflux disease (GERD)-related esophageal (heartburn and regurgitation) and extra-esophageal symptoms, heal esophagitis and reduce hiatal hernia; (II) the capability to control gastro-esophageal reflux, as measured by 24-h pH-impedance recording; (III) the long-term efficacy of the procedure, compared with surgery; (IV) the cost-effectiveness of the procedure, compared with surgery.
Efficacy of TIF in controlling GERD symptoms
Two meta-analyses, published in 2017 and 2018, assessed the outcomes of TIF.
The first meta-analysis assessed only the outcomes of TIF performed by EsophyX and included 18 studies, 13 prospective observational studies and 5 randomized controlled trials, published between 2007 and 2015 and evaluating 963 patients (5). Among the 13 observational studies, two provided results in 3 months, nine in 6 months, seven in 12 months, three in 24 and 36 months, and one in 4, 5, and 6 years. Symptoms outcomes included the patient satisfaction rate, defined as an improvement of at least 50% in the GERD health-related quality of life (GERD-HRQL) scores, remission of heartburn and regurgitation, complete cessation or reduction of PPIs usage, esophageal acid exposure time (% time pH <4), 24-h total number of refluxes, and 24-h acid reflux episodes.
Cessation of PPI consumption was reported in 31.6% to 82% of cases; cessation or reduction by 50% of PPI consumption was reported in 63% to 89% of cases, with an average rate ranging between 80% and 89% over a 6- to 72-month follow-up. The overall satisfaction rate varied from 58% to 86%, with a weighted average rate of 69.15%. In four randomized controlled trials the pooled relative risk of response rate to TIF versus PPIs/sham was 2.44 (95% CI: 1.25–4.79, P=0.0009) in the intention-to-treat analysis (6-9).
A more recent meta-analysis including both EsophyX and MUSE studies has been published in 2018 (10). Compared with the previous meta-analysis, this study analyzed more published papers [32] and patients [1,475], and considered as outcomes the post-procedure occurrence of typical and atypical GERD symptoms, measured as GERD health-related quality of life (GERD-HRQL), gastroesophageal reflux symptom score (GERSS), and reflux symptom index (RSI), the cessation of PPI consumption rather than reduction, and functional findings when available (esophageal acid exposure time, number of reflux episodes, and De-Meester scores). Overall, TIF has proven to be technically feasible with an immediate success rate of 99% and with a 2% rate of serious adverse events. Mean GERD HRQL significantly improved after TIF compared with pre-procedure scores (mean difference 17.72, P<0.001); mean GERSS was significantly lower post-procedure (mean difference 23.78, P<0.001); mean RSI was also significantly lower post-procedure (mean difference 14.28, P<0.001). Complete discontinuation of PPI therapy was achieved post-procedure in a significant number of patients (89%, P<0.001).
Both meta-analyses concluded that TIF is an effective alternative to surgery to control GERD-related symptoms with comparable short-term patient satisfaction.
Only three studies assessed so far the outcomes after TIF performed by MUSE technique. A pilot study assessed GERD-related symptoms and PPI use up to 5 years after the procedure in 13 subjects: GERD-related symptom score at six month was normalized in 92% of cases, PPI use was completely stopped or reduced by half in 77% of cases (54% off PPI completely). PPI therapy was abolished or reduced by half in 82% of patients at 12 months and in 73% at 36 months. This rate persisted unchanged up to 5 years (11).
In a multicenter, prospective international study enrolling 66 patients with a 6-month follow-up, GERD-related symptoms scores improved by more than 50% in 73% of patients, and 85% were no longer using PPI or markedly reduced by more than 50% the daily dose; 64.6% of patients discontinued PPI medication. At 24-hour pH-recording the total time with esophageal pH <4.0 decreased significantly from baseline (12).
Another study assessed efficacy and safety of TIF performed by MUSE in 37 patients at baseline, 6 months, and up to 4 years post-procedure in a single center (13). The proportions of patients who remained off daily PPI were 83.8% (31/37) at 6 months and 69.4% (25/36) at 4 years post-procedure. GERD-HRQL scores (off PPI) were significantly decreased from baseline to 6 months and 4 years post-procedure. The daily dosage of GERD medications, measured as omeprazole equivalents (mean ± SD, mg), decreased from 66.1±33.2 at baseline to 10.8±15.9 at 6 months and 12.8±19.4 at 4 years post-procedure (P<0.01).
Regurgitation is less responsive to acid suppression than heartburn in GERD patients and is a common cause of treatment failure (14). In a study including 87 patients undergone TIF and then 6-month placebo and 42 patients undergone sham surgery and then 6-month PPI therapy, at the intention-to-treat analysis TIF and PPIs eliminated troublesome regurgitation in 67% and 45% of patients, respectively (P=0.02) (6).
In the TEMPO randomized trial with a crossover arm, elimination of regurgitation after TIF by EsophyX was achieved in 88% of cases at 6 months and 1 year, 90% of cases at 2 and 3 years, and 86% at 5 years; atypical GERD symptoms were eliminated in 88% of patients (7,15,16).
Extra-esophageal symptoms are the most difficult to cure, compared with esophageal ones; in general high doses of PPIs are required for a long time (up to 6 months) to achieve clinical remission of symptoms. In the TEMPO study extra-esophageal symptoms were significantly reduced both at 6 months and 1 year, 2 and 3, and 5 years. Resolution of troublesome atypical symptoms was achieved in 82% of patients at 1 year, 88% at 3 years, and 80% at 5 years (7,15,16).
As it regards the effect of TIF on hiatal hernia, reduction or complete resolution has been achieved in 91% of patients (P<0.001) in the meta-analysis published in 2018 (10).
Efficacy of TIF in controlling gastro-esophageal reflux
In the meta-analysis published in 2017 on Esophyhx procedures, 11 studies evaluated pre- and post-procedure pH +/− impedance recordings. Five RCTs evaluated the esophageal acid exposure time before and after TIF procedure. TIF showed similar efficacy with respect to esophageal acid exposure time compared with PPIs and improved patients’ acid exposure time compared with sham groups (6,7). Three RCTs (6,8,17) reported the total reflux episodes before and after TIF therapy: patients undergoing TIF yielded significant reduction in reflux episodes compared with those who did not (P<0.00001). Two RCTs (17,18) reported the incidence of acid reflux episodes before and after TIF: patients undergoing TIF showed no significant differences from those who received PPIs therapy (P=0.16).
In the meta-analysis published in 2018, esophageal acid exposure time (i.e., percent time with pH <4) was reported in 15 studies (n=722) and significantly improved after the TIF intervention (P<0.001). The number of reflux episodes in a 24-hour period also significantly improved from pre-procedure levels (P<0.001). Post-procedure DeMeester scores were reported in 11 studies (n=647) and improved significantly (P<0.001), too.
Efficacy of TIF on long-term follow-up
Most of studies included in the two meta-analyses report outcomes at one or two years, so the long-term efficacy of TIF still remains an open issue. As it regards the duration of the symptom control after TIF, 7 studies reported outcomes at 3 to 6 years after the intervention, performed either by EsophyX (5 studies) or MUSE (2 studies). At 3-year follow-up, discontinuation of daily PPI ranged from 74% to 84% of cases (15,19,20). At 4, 5 and 6 years, complete cessation of PPI usage ranged from 35.7% to 73% of cases (11,13,16,20,21). In the Tempo trial (16), at the 5-year follow-up 34% of patients were on daily PPI therapy as compared with 100% of patients at screening; the total GERD-HRQL score improved by decreasing from 22.2 to 6.8 (P<0.001).
Data are summarized in Table 1.
Table 1
| Study | Device | Number of patients | Number of centres | Follow-up duration (years) | % patients off PPIs | % patients off or halved PPIs |
|---|---|---|---|---|---|---|
| Roy-Shapira, |
MUSE | 13 | 1 | 5 | 64 | 77 |
| Testoni |
EsophyX | 14 | 1 | 6 | 35.7 | 85.7 |
| Kim |
MUSE | 37 | 3 | 4 | 54 | – |
| Stefanidis |
EsophyX | 44 | 1 | 5 | 73 | 86 |
| Trad |
EsophyX | 44 | 1 | 5 | 46 | 66 |
TIF, transoral incisionless fundoplication; MUSE, medigus ultrasonic surgical endostapler; PPI, proton pump inhibitor.
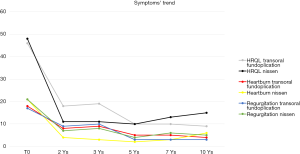
Outcomes up to 10 years after TIF by EsophyX have been assessed only by our group. Over the 10-year follow-up the GERD-HRQL scores off PPI therapy and heartburn and regurgitation scores remained significantly lower than the pre-TIF scores. The reduction was significant at 2 years and scores were similar at 3 years; between 3 and 5 years there was a further slight reduction in the mean scores; at 7 and 10 years scores did not substantially change from the 5-year picture. Mean GERD-HRQL, heartburn, and regurgitation scores appeared substantially unchanged between 2 and 10 years after the TIF procedure. The percentages of patients who had stopped or halved the PPI therapy 2, 3, 5, 7 and 10 years after TIF were respectively 86.7%, 84.4%, 73.5%, 83.3%, and 91.7%; the rates remained substantially stable and similar to that at 2 years, with a tendency toward improvement at 10 years. Considering as responders to TIF only patients who completely stopped PPI therapy, there were respectively 55.6%, 53.3%, 41.2%, 45.8%, and 41.7% full responders at 2, 3, 5, 7 and 10 years. The complete response rate after TIF had fallen by about 20% at 10 years compared to that at 2 years but the differences were not significant (P>0.05) (Figure 1). Although only a relatively small number of patients had a 10-year post-TIF follow-up, the symptomatic curve over that period suggests that the results would not change much even with a larger number of cases.
Long-term efficacy of TIF has been reported also for regurgitation and atypical symptoms. In the TEMPO trial, troublesome regurgitation was eliminated in 90% of patients at 3 years and 86% at 5 years; atypical symptoms in 88% of patients at 3 years and 80% at 5 years (16).
Results of our series and TEMPO trial demonstrate that factors that negatively affect outcomes play a role only in the early post-operative period; patients who still respond 1 to 2 years after the operation enjoy a substantially unchanged response over the next few years.
In our published series (20), pre-operative Hill grades III and IV and hiatal hernia larger than 2 cm at endoscopy, a small number of fasteners deployed (median 10±2), and the presence of severe ineffective esophageal motility at manometry were all associated with a higher rate of early unsuccessful results. The deployment of a larger number of fasteners (median 14±2) raised the probability of being a responder about four-fold, as stated in another paper (22). The adoption of the rotational technique to create the new valve gave a 50% increase in successful outcomes too, according to other studies (22,23).
Ineffective esophageal motility is a problem seen in a heterogeneous group with different manometric subsets and different symptom profiles, who might respond to the procedure in different ways (24).
Twenty-one studies including 1,176 patients specifically addressed the need for repeat endoscopic procedure or alternative surgery secondary to poorly controlled GERD symptoms (10). A total of 7.5% patients required further endoscopic or surgical intervention; the majority (89.5%) of these revisions were performed within 6 months of the TIF procedure, confirming again that failure of TIF occurs in general during the first year after the intervention. Only three studies (9 patients in total) assessed for symptomatic improvement after repeat endoscopic or surgical intervention; 7 of the 9 patients (77.8%) reported symptom improvement.
Two- to three-year post-TIF outcomes are similar to those with surgical posterior partial (Toupet) or anterior partial fundoplication, but inferior to those reported in patients operated by posterior total fundoplication (Nissen) (25,26), though without any of the surgery-related persistent side effects. On the other hand, 5-year post-TIF results were substantially similar to those reported with Nissen fundoplication. A nationwide register-based follow-up study on the use of PPI after anti-reflux surgery reported that at 5 years 57.5% and 29.5% of patients still took PPI or were daily PPI-dependent (27). In the Cochrane meta-analysis on Nissen fundoplication the pooled analysis of long-term results showed recurrence or persistence of heartburn and reflux symptoms in 41.2% and 24.6% of cases, respectively, with persistent side effects in 14% up to 23% of cases (26).
There are few reports of 10-year outcomes after surgical fundoplication but, again, results are substantially comparable with our TIF findings (27-29). In a systematic review in partial responders to PPI who had undergone laparoscopic fundoplication, 10 years after the operation 35.8% reported heartburn and 29.1% regurgitation, with an 18.2% rate of acid-suppressive medication (28). The nationwide register-based follow-up study on the use of PPI after antireflux surgery showed that at ten years 72.4% and 41.1% of patients were taking PPI or were daily PPI-dependent (27).
Cost-effectiveness of TIF compared with laparoscopic Nissen fundoplication
An important issue regarding the introduction in clinical practice of a new therapeutic approach alternative to surgery is the cost-effectiveness compared with the standard approach (in this case laparoscopic Nissen fundoplication). The issue is relevant considering the huge number of potential patients asking for the TIF procedure in alternative to surgery. We can also argue that patients asking for TIF represent a different group not comparable with those referred to laparoscopic Nissen fundoplication.
To date, only the TEMPO study aimed at assessing the cost-effectiveness of TIF versus LNF (16). Authors found in the Optum database, 2,734 LNF patients and 73 TIF patients available for the cost analysis. The average individual cost and health care utilization over 2 years for a patient undergone LNF was $99,256, as compared with $71,691 for a TIF patient. In the subgroup of most refractory PPI patients the average cost of care over a 2-year period was for a LNF patient $124,000, as compared with $66,000 for a TIF patient.
Conclusions
In conclusion, TIF performed either by EsophyX or MUSE has been shown as effective as surgery in controlling not only heartburn but also regurgitation and atypical GERD symptoms which are less responsive to PPI treatment compared with heartburn, without any of the surgery-related persistent side effects such as dysphagia and gas bloat.
Although the two available meta-analyses showed that patients tend to resume PPI therapy at reduced dose at 12–24 months follow-up, 2- to 3-year outcomes are substantially similar to those reported for anterior surgical fundoplication, but worse than for total fundoplication; 5- and 10-year outcomes are very similar to those for Nissen fundoplication. As it regards the cost analysis, TIF seems to be more cost-effective than LNP.
To date available findings are sufficient to confirm that TIF appears to offer an effective and safe therapeutic option for carefully selected symptomatic GERD patients, with Hill grade of the valve I and II or hiatal hernia not longer than 2 cm, who refuse life-long medical therapy or surgery, are intolerant to PPI, or have some risk of persistent post-surgical side effects.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editor (Luigi Bonavina) for the series “Gastroesophageal Reflux Disease” published in Annals of Esophagus. The article has undergone external peer review.
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aoe.2018.08.01). The series “Gastroesophageal Reflux Disease” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Cadière GB, Rajan A, Germay O, et al. Endoluminal fundoplication by a transoral device for the treatment of GERD: a feasibility study. Surg Endosc 2008;22:333-42. [Crossref] [PubMed]
- Kauer WK, Roy-Shapira A, Watson D, et al. Preclinical trial of a modified gastroscope that performs a true anterior fundoplication for the endoluminal treatment of gastroesophageal reflux disease. Surg Endosc 2009;23:2728-31. [Crossref] [PubMed]
- Cadière GB, Van Sante N, Graves JE, et al. Two-year results of a feasibility study on antireflux transoral incisionless fundoplication using Esophyx. Surg Endosc 2009;23:957-64. [Crossref] [PubMed]
- Hoppo T, Immanuel A, Schuchert M, et al. Transoral incisionless fundoplication 2.0 procedure using EsophyX™ for gastroesophageal reflux disease. J Gastrointest Surg 2010;14:1895-901. [Crossref] [PubMed]
- Huang X, Chen S, Zhao H, et al. Efficacy of transoral incisionless fundoplication (TIF) for the treatment of GERD: a systematic review with meta-analysis. Surg Endosc 2017;31:1032-44. [Crossref] [PubMed]
- Hunter GJ, Kahrilas PJ, Bell RC, et al. Efficacy of transoral fundoplication vs omeprazole for treatment of regurgitation in a randomized controlled trial. Gastroenterology 2015;148:324-333.e5. [Crossref] [PubMed]
- Trad KS, Barnes WE, Simoni G, et al. Transoral incisionless fundoplication effective in eliminating GERD symptoms in partial responders to proton pump inhibitor therapy at 6 months: the TEMPO randomized clinical trial. Surg Innov 2015;22:26-40. [Crossref] [PubMed]
- Håkansson B, Montgomery M, Cadiere GB, et al. Randomised clinical trial: transoral incisionless fundoplication vs. sham intervention to control chronic GERD. Aliment Pharmacol Ther 2015;42:1261-70. [Crossref] [PubMed]
- Witteman BP, Conchillo JM, Rinsma NF, et al. Randomized controlled trial of transoral incisionless fundoplication vs. proton pump inhibitors for treatment of gastro-esophageal reflux disease. Am J Gastroenterol 2015;110:531-42. [Crossref] [PubMed]
- McCarty TR, Itidiare M, Njei B, et al. Efficacy of transoral incisionless fundoplication for refractory gastroesophageal reflux disease: a systematic review and meta-analysis. Endoscopy 2018;50:708-25. [Crossref] [PubMed]
- Roy-Shapira A, Bapaye A, Date S, et al. Trans-oral anterior fundoplication: 5-year follow-up of pilot study. Surg Endosc 2015;29:3717-21. [Crossref] [PubMed]
- Zacherl J, Roy-Shapira A, Bonavina L, et al. Endoscopic anterior fundoplication with the Medigus Ultrasonic Surgical Endostapler (MUSE) for gastroesophageal reflux: 6-month results from a multi-center prospective trial. Surg Endosc 2015;29:220-9. [Crossref] [PubMed]
- Kim HJ, Kwon CI, Kessler WR, et al. Long-term follow-up results of endoscopic treatment of gastroesophageal reflux disease with the MUSE™ endoscopic stapling device. Surg Endosc 2016;30:3402-8. [Crossref] [PubMed]
- Kahrilas PJ, Jonsson A, Denison H, et al. Regurgitation is less responsive to acid suppression than heartburn in patients with gastroesophageal reflux disease. Clin Gastroenterol Hepatol 2012;10:612-9. [Crossref] [PubMed]
- Trad KS, Fox MA, Simoni G, et al. Transoral fundoplication offers durable symptom control for chronic GERD: 3-year report from the TEMPO randomized trial with a crossover arm. Surg Endosc 2017;31:2498-508. [Crossref] [PubMed]
- Trad KS, Barnes WE, Prevou ER, et al. The TEMPO Trial at 5 Years: Transoral Fundoplication (TIF 2.0) is safe, durable, and cost-effective. Surg Innov 2018;25:149-57. [Crossref] [PubMed]
- Rinsma NF, Farre R, Bouvy ND, et al. The effect of endoscopic fundoplication and proton pump inhibitors on baseline impedance and heartburn severity in GERD patients. Neurogastroenterol Motil 2015;27:220-8. [Crossref] [PubMed]
- Trad KS, Simoni G, Barnes WE, et al. Efficacy of transoral fundoplication for treatment of chronic gastroesophageal reflux disease incompletely controlled with high-dose proton-pump inhibitors therapy: a randomized, multicenter, open label, crossover study. BMC Gastroenterol 2014;14:174. [Crossref] [PubMed]
- Muls V, Eckardt AJ, Marchese M, et al. Three-year results of a multicenter prospective study of transoral incisionless fundoplication. Surg Innov 2013;20:321-30. [Crossref] [PubMed]
- Testoni PA, Testoni S, Mazzoleni G, et al. Long term efficacy of transoral in cionless fundoplication with Esophyx (TIF 2.0) and factors affecting outcomes in GERD patients followed for up to 6 years: a prospective single-center study. Surg Endosc 2015;29:2770-80. [Crossref] [PubMed]
- Stefanidis G, Viazis N, Kotsikoros N, et al. Long-term benefit of transoral incisionless fundoplication using the Esophyx device for the management of gastroesophageal reflux disease responsive to medical therapy. Dis Esophagus 2017;30:1-8. [PubMed]
- Bell RC, Cadière GB. Transoral rotational esophagogastric fundoplication: technical, anatomical, and safety considerations. Surg Endosc 2011;25:2387-99. [Crossref] [PubMed]
- Bell RC, Mavrelis PG, Barnes WE, et al. A prospective multicenter registry of patients with chronic gastro-esophageal reflux disease receiving transoral incisionless fundoplication. J Am Coll Surg 2012;215:794-809. [Crossref] [PubMed]
- Haack HG, Hansen RD, Malcolm A, et al. Ineffective esophageal motility: manometric subsets exhibit different symptom profiles. World J Gastroenterol 2008;14:3719-24. [Crossref] [PubMed]
- Broeders JA, Mauritz FA, Ahmed Ali U, et al. Systematic review and meta-analysis of laparoscopic Nissen (posterior total) versus Toupet (posterior partial) fundoplication for gastro-esophageal reflux disease. Br J Surg 2010;97:1318-30. [Crossref] [PubMed]
- Garg SK, Gurusamy KS. Laparoscopic fundoplication surgery versus medical management for gastro-oesophageal reflux disease (GORD) in adults. Cochrane Database Syst Rev 2015;CD003243 [PubMed]
- Lødrup A, Pottegard A, Hallas J, et al. Use of proton pump inhibitors after antireflux surgery. A nationwide register-based follow-up study. Gut 2014;63:1544-9. [Crossref] [PubMed]
- Lundell L, Bell M, Ruth M. Systematic review: laparoscopic fundoplication for gastro-esophageal reflux disease in partial responders to proton pump inhibitors. World J Gastroenterol 2014;20:804-13. [Crossref] [PubMed]
- Kelly JJ, Watson DI, Chin KF, et al. Laparoscopic Nissen fundoplication: clinical outcomes at 10 years. J Am Coll Surg 2007;205:570-5. [Crossref] [PubMed]
Cite this article as: Testoni PA, Testoni S. Transoral fundoplication for gastroesophageal reflux disease. Ann Esophagus 2018;1:7.


