The upper esophageal sphincter: anatomy and physiology
Introduction
In humans, the reality of a combined upper aerodigestive tract necessitates the finely tuned regulation of the division into the digestive and respiratory systems. The larynx and vocal folds represent the entry into the airway, while the upper esophageal sphincter (UES) guards the entrance of the esophagus, both from aerophagia in the anterograde direction and regurgitation in the retrograde (1). During the act of swallow, the relaxation of the cricopharyngeus muscle (CPM), elevation of the larynx and propulsion of the food bolus by the pharynx coordinate to open the UES and facilitate deglutition (2). A thorough understanding of the UES anatomy and physiology is critical for all deglutologists seeking to improve pharyngo-esophageal swallow function.
Methods
Given the scope of the material, a narrative review format was chosen. Prior material based on the senior author’s (PB) experience and scholarship in addition to relevant recent publications on the relevant topics of anatomy and physiology.
Anatomy
Terminology
There are several terms that are often used in discussion of the UES. These are the CPM, the pharyngoesophageal segment (PES), and the upper esophageal high-pressure zone (UEHPZ). A review of terminology is a necessary foundation for a comprehensive understanding of UES anatomy and physiology (3). The word sphincter is defined as an annular muscle surrounding and able to contract or close a bodily opening (4). No aspect of the UES entirely meets this definition. Instead of being a true sphincter, the UES is a 4-cm segment of the digestive tract consisting of cartilaginous, bony and muscular components that opens and closes based on both intrinsic muscular control and displacement. The PES is synonymous with this definition of the UES. The CPM, however, is not synonymous with the UES and PES. Although the CPM is an important component of the UES, it only represents the distal third of the UES. The UEHPZ is a manometric term referring to the areas of high pressure noted in the anatomic region of the UES.
Components of the UES
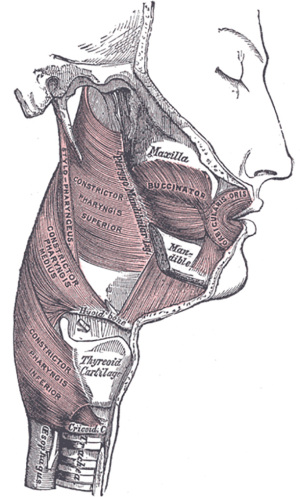
The UES is a kidney bean shaped potential space, about 4 cm in length, that is bounded anteriorly by the larynx, posterolaterally by the pharyngoesophageal muscles, superiorly by the pharynx and inferiorly by the esophagus (5-7). The bulk of the pressure providing the sphincteric function of the region is from the elastic recoil of the larynx abutting the cervical spine. The muscular control of the UES is primarily extrinsic by the suprahyoid and infrahyoid muscles, while intrinsic control of opening and closing is by the CPM, inferior constrictor muscle and the cranial circular esophageal muscle.
The larynx
The larynx is the most highly protected region of the upper aerodigestive tract. A major purpose of the coordinated swallow is to prevent penetration and aspiration into the larynx and trachea and allow appropriate passage through the UES into the esophagus (1). The posterior aspect of the larynx, including the arytenoid and cricoid cartilages also forms the anterior wall of the UES. The inferior constrictor muscles attach to the thyroid laminae, while the CPM attaches to the posterolateral aspects of the cricoid cartilage. At rest, these cartilages press against the spine, closing the potential space of the UES. During deglutition, the larynx is elevated off the spine by cervical and pharyngeal muscles decreasing the pressure required by the pharynx to push a food bolus into the UES and to the esophagus beyond.
The extrinsic muscles of the UES
As stated above, the primary source of closing pressure on the UES is that of the larynx pressing the UES closed against the spine. Thus, the release of this pressure by elevation of the larynx is critical for opening of the UES. The extrinsic muscles controlling UES function are divided into anterior and posterior groups. The anterior group consists of the suprahyoid (geniohyoid, mylohyoid, stylohyoid, hyoglossus and anterior belly of digastric) and infrahyoid muscles (thyrohyoid, sternohyoid, sternothyroid and omohyoid). The suprahyoid muscles move the hyoid bone anterosuperiorly with contraction while the thyrohyoid and omohyoid pull the larynx up and anteriorly with the hyoid bone. The remaining infrahyoid muscles stabilize the larynx during deglutition. The posterior group of extrinsic UES opening muscles include the stylopharyngeus, palatopharyngeus, and pterygopharyngeus. These muscles shorten the pharynx, pulling the UES upwards while concomitantly widening it. The opening muscles are primarily composed of fast-twitch fibers, with the exception of the thyrohyoid muscle, which consists of 40% slow-twitch fibers, suggestive of increased fatigue resistance necessary for a tonic role of the muscle. This is not unexpected as the thyrohyoid serves to maintain a stable distance between the larynx and the hyoid bone.
The intrinsic muscles of the UES
The intrinsic muscles of the UES provide additional sphincteric function and propulsive force for a food bolus. The primary intrinsic muscle is the CPM, while the inferior constrictor and cranial aspect of the circular esophageal muscle are also involved. The inferior constrictor and CPM are attached anteriorly to the posterolateral aspects of the thyroid and cricoid cartilages respectively. See Figure 1 for an illustration of the muscular anatomy of the pharynx and UES. The circular esophageal muscle is not attached to the larynx. Together, these form a pliable and elastic tube posterior to the larynx that can expand and contract passively with a food bolus in addition to actively contracting to prevent the swallowing of air during respiration and phonation and the regurgitation of esophageal contents into the airway. It is this phenomenon of two nesting semicircles, the muscular one outer and the cartilaginous one inner, that creates the kidney bean shape of the UES as demonstrated by 3D reconstructions and cadaveric studies (6,7).
CPM
Representing the caudal third of the UES, the CPM is the primary intrinsic muscular sphincter within the UES. The CPM is a striated muscle with horizontally oriented (CPh/pars fundiformis) and obliquely oriented fibers (CPo/pars obliqua). The muscle consists of both fast and slow twitch muscle fibers, though slow twitch fibers predominate (69–76%), speaking to its role in basal tone for the UES in addition to more rapid functions (8). The CPM contains more elastic connective tissue elements than other muscles of the pharynx. This allows the CP to stretch up to 1.7 times its basal length prior to exerting maximum active tension. This further contributes to the passive recoil of the UES after passage of a food bolus (9,10).
Inferior constrictor muscle and cranial circular esophageal muscle
The inferior constrictor muscle and the cranial circular esophageal muscles have also been found to serve roles in UES function. Although not always activated, they have demonstrated activity during respiration, retching and vomiting. The IPC in particular has been sown to be capable of exerting more force at maximum contraction compared to the CPM, while the proximal cervical esophagus has an innervation pattern similar to other digestive sphincters.
The cervical spine
The UES is typically located at C5–C6 at rest along the cervical spine with the vertebral bodies comprising the posterior wall of the UES with a thin layer of intervening muscles and mucosa (11). Changes in the curvature of the spine in addition to degenerative changes of the cervical spine such as osteophytes can change the 3D geometry of the UES. Additionally, surgical changes such as anterior cervical spine plates and screws can impinge on the UES, potentially even causing perforation.
Innervation
The CP is bilaterally innervated with motor innervation supplied primarily from the pharyngoesophageal nerve, a branch of the vagus nerve. The inferior pharyngeal constrictor is also primarily supplied by the pharyngeal branch of the vagus through the pharyngeal plexus, while the cervical esophagus is innervated by the recurrent laryngeal nerve. Sensory innervation of the mucosa and musculature are from the glossopharyngeal nerve, the internal branch of the superior laryngeal nerve, and the vagus nerve. The area is able to sense fine touch, deep touch, stretch, temperature and pain (8,12).
UES physiology
UES activity during normal swallow
The normal swallow consists of 5 phases of UES activity. Two phases are due to active opening and closing of the CPM and the remaining three are due to passive effects of normal swallow physiology. These five phases are (I) muscular relaxation of the CPM, (II) elevation of the larynx, (III) distention of the UES by the intrabolus pressure exerted upon the advancing bolus, (IV) passive closure of the UES by elastic recoil of the larynx against the spine, and (V) active closure through CPM contraction (3,13).
Phase 1: muscular relaxation of the CPM
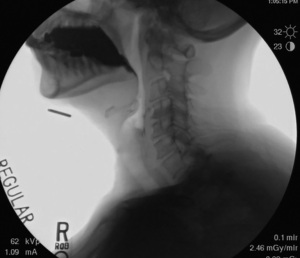
The first step of UES opening is relaxation of the CPM. The tonic contraction of the CPM is relaxed due to inhibited input from the vagus nerve. This inhibition precedes the opening of the UES by approximately 100–200 ms (14). During deglutition the CPM is relaxed for about 0.3–0.6 seconds in humans depending on bolus characteristics. Throughout phase 1, the larynx remains low in the neck and the UES remains closed. See Figure 2 for the fluoroscopic appearance of the larynx during phase 1 with the bolus in the oral cavity and the larynx low in the neck.
Phase 2: elevation of the larynx
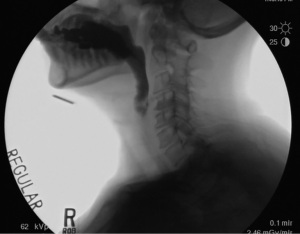
During phase 2, the anterior and posterior groups of the UES opening muscles contract to elevate the larynx, bringing it anteriorly under the tongue base to expand the hypopharyngeal space and to prime the UES for opening. At the same time the infrahyoid muscles are completely relaxed allowing maximum movement of the hyoid, larynx and UES. This elevation can be seen in Figure 3. This phase has been described as a passive phase that depends on the movement of other structures in the swallowing system. However, a recent histological study shows that the palatopharyngeus muscle is connected to the cricopharyngeal part of the inferior constrictor muscle through a dense connective tissue (15). The article’s authors suggest that the palatopharyngeus may act directly on the UES-opening by elevating the cricopharyngeal muscle superolaterally in addition to the superior-anterior movement of the hyolaryngeal complex. The initial opening of the UES due to the traction of the hyolaryngeal complex is approximately 7 mm (16,17). It is the movement of the hyolaryngeal complex that determines the distention of UES opening for dry swallows and small bolus volumes. However, it is phase 3 that results in true opening of the UES after priming by CPM relaxation and hyolaryngeal elevation.
Phase 3: PES distention
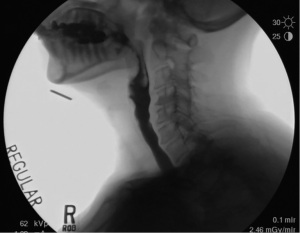
The distention of the UES is adaptive to bolus volume, viscosity and intrabolus pressure. The elasticity of the UES allows it to expand further with increasing bolus volume or heavier viscosity. Intrabolus pressure also plays an important role on the distention of the UES, regulated by lingual and pharyngeal peristalsis to propel the bolus through the hypopharynx into the expanding UES. Normal values of UES distention range from approximately 5.6–14.0 mm. Maximal UES distention occurs approximately 70–170 ms after initial UES opening (17). The maximal opening of the UES can be noted in Figure 4.
Phase 4: passive closure through elastic recoil
After maximal distention is reached, the UES starts to collapse as an effect of elastic recoil in addition to relaxation of the suprahyoid muscles and return of the hyolaryngeal complex to its basal lower position. Figure 5 demonstrates phase 4 of the UES opening with the bolus in esophagus while the larynx has returned to its resting position.
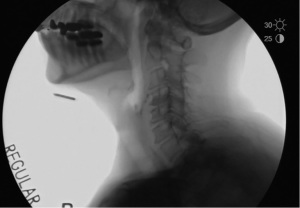
Phase 5: active closure through CPM contraction
The last phase of UES activity is the reactivation of contraction of the CPM, which occurs as the pharyngeal wave hits the sphincter (Figure 6).
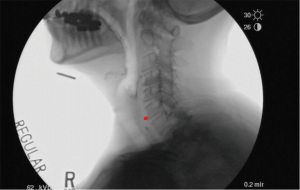
Conclusions
The UES serves to protect the esophagus and stomach from aerophagia and the larynx from gastric and esophageal reflux while facilitating normal swallow function in concert with the suprahyoid muscles, pharynx, larynx and esophagus. An understanding of the anatomy of the region in addition to the phases of UES opening facilitates more refined therapeutic interventions for patients with pharyngoesophageal swallow impairment and dysphagia.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editor (Fernando A. M. Herbella) for the series “Upper Esophageal Sphincter” published in Annals of Esophagus. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://aoe.amegroups.com/article/view/10.21037/aoe-21-34/coif). The series “Upper Esophageal Sphincter” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Sasaki CT. Laryngeal Physiology for the Surgeon and Clinician. Second Edition ed. San Diego, CA: Plural Publishing, 2017.
- Cook IJ, Dodds WJ, Dantas RO, et al. Opening mechanisms of the human upper esophageal sphincter. Am J Physiol 1989;257:G748-59. [PubMed]
- Belafsky PC. Clinical Esophagology and Transnasal Esophagoscopy. San Diego, CA: Plural Publishing, 2019.
- "Sphincter." Merriam-Webster.com. Available online: https://www.merriam-webster.com/dictionary/sphincter
- Randall DR, Cates DJ, Strong EB, et al. Three-dimensional analysis of the human pharyngoesophageal sphincter. Laryngoscope 2020;130:2773-8. [Crossref] [PubMed]
- Belafsky PC, Plowman EK, Mehdizadeh O, et al. The upper esophageal sphincter is not round: a pilot study evaluating a novel, physiology-based approach to upper esophageal sphincter dilation. Ann Otol Rhinol Laryngol 2013;122:217-21. [Crossref] [PubMed]
- Cates D, Plowman EK, Mehdizadeh O, et al. Geometric morphometric shape analysis in an ovine model confirms that the upper esophageal sphincter is not round. Laryngoscope 2013;123:721-6. [Crossref] [PubMed]
- Lang IM. Upper Esophageal Sphincter. 2006. In: GI Motility Online [Internet]. Nature Publishing Group. Available online: https://www.nature.com/gimo/contents/pt1/full/gimo12.html#relatedcontent
- Asoh R, Goyal RK. Manometry and electromyography of the upper esophageal sphincter in the opossum. Gastroenterology 1978;74:514-20. [Crossref] [PubMed]
- Bonington A, Mahon M, Whitmore I. A histological and histochemical study of the cricopharyngeus muscle in man. J Anat 1988;156:27-37. [PubMed]
- Abdel-Aziz M, Azab N, El-Badrawy A. Cervical osteophytosis and spine posture: contribution to swallow disorders and symptoms. Curr Opin Otolaryngol Head Neck Surg 2018;26:375-81. [Crossref] [PubMed]
- Lang IM, Shaker R. An overview of the upper esophageal sphincter. Curr Gastroenterol Rep 2000;2:185-90. [Crossref] [PubMed]
- Jacob P, Kahrilas PJ, Logemann JA, et al. Upper esophageal sphincter opening and modulation during swallowing. Gastroenterology 1989;97:1469-78. [Crossref] [PubMed]
- Kendall KA, McKenzie S, Leonard RJ, et al. Timing of events in normal swallowing: a videofluoroscopic study. Dysphagia 2000;15:74-83. [Crossref] [PubMed]
- Fukino K, Tsutsumi M, Nimura A, et al. Anatomy of inferior end of palatopharyngeus: its contribution to upper esophageal sphincter opening. Eur Arch Otorhinolaryngol 2021;278:749-54. [Crossref] [PubMed]
- Regan J, Walshe M, Rommel N, et al. New measures of upper esophageal sphincter distensibility and opening patterns during swallowing in healthy subjects using EndoFLIP®. Neurogastroenterol Motil 2013;25:e25-34. [Crossref] [PubMed]
- Ferris L, Doeltgen S, Cock C, et al. Modulation of pharyngeal swallowing by bolus volume and viscosity. Am J Physiol Gastrointest Liver Physiol 2021;320:G43-53. [Crossref] [PubMed]
Cite this article as: Ramaswamy AT, Martell P, Azevedo R, Belafsky P. The upper esophageal sphincter: anatomy and physiology. Ann Esophagus 2022;5:30.