Minimally invasive transhiatal esophagectomy
Introduction
Esophagectomy is used for the treatment of esophageal cancer as well as benign disease. Transhiatal esophagectomy with a cervical anastomosis was reported by Orringer et al. in 1978 (1) and developed into a safe, reproducible, oncologically sound procedure (2,3). The advantages compared to other types of esophagectomies are the avoidance of thoracic incisions, which decreases respiratory complications; and a cervical anastomosis, which is less morbid if it leaks. The main criticism is an incomplete thoracic lymph node dissection, since the mediastinal dissection is performed bluntly. There is no clear difference in oncologic outcomes when comparing the transhiatal esophagectomy to other types.
The first robotic transhiatal esophagectomy was reported by Horgan et al. in 2003, with a robot-assisted abdominal portion and an open cervical portion (4). Initial case series from several institutions showed good surgical and oncologic outcomes using similar approaches (5-7). The video-assisted cervical approach was first described by Bumm et al. in 1993 and allowed a more complete thoracic lymph node dissection. The techniques have been refined using smaller scopes and insufflation (8,9). Transhiatal esophagectomy using combinations of video- and robot-assisted abdominal and cervical approaches (10,11), as well as robot-assisted approaches for both the abdominal and cervical portions, have been described (12,13). Most surgeons favor a small laparotomy to extract the specimen.
In this article, one of the authors (DS Demos) shares his technique in performing a minimally invasive transhiatal esophagectomy, with a robot-assisted abdominal and video-assisted cervical approach. The main advantage of the robot-assisted abdominal approach is the magnified, stereotactic view provided by the camera and the increased dexterity provided by the instruments. Using a camera in both the abdominal and cervical portions of the procedure allows a complete thoracic lymph node dissection.
Preoperative planning
Patients undergo extensive preoperative evaluation with upper endoscopy; computed tomography (CT) scan of the chest, abdomen, and pelvis with intravenous (IV) contrast; fluorodeoxyglucose-positron-emission tomography/CT (FDG-PET/CT) evaluation; endoscopic ultrasound if endoscopic or surgical resection alone may be potential treatment; and bronchoscopy if the tumor is near the airway. Patients who have had preoperative chemoradiation should undergo restaging FDG-PET/CT to ensure that their disease is still resectable. Patients with malnutrition may undergo feeding jejunostomy before neoadjuvant therapy.
Anesthesia and positioning
General anesthesia is induced with single-lumen orotracheal intubation. Only peripheral IV access is needed. Central access, if necessary for any reason, can be established in the right internal jugular vein or either groin. Arterial line placement is not routinely necessary. Upper endoscopy is performed to confirm the location of the tumor. The patient is placed supine with both arms tucked. The neck is extended and rotated to the right to provide access to the left neck.
General conduct
Both the abdominal and cervical portions of this procedure can be performed simultaneously. Operative time is reduced substantially and anesthetic delivery is minimized. This provides for a less-eventful initial postoperative period when perfusion of the conduit and anastomosis is critical.
Robot-assisted abdominal portion
Port placement and docking
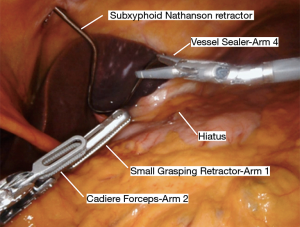
Port placement for this portion of the procedure is generally acceptable for all foregut operations. It utilizes all 8-mm ports, essentially in the same transverse plane. We prefer blunt needle abdominal insufflation just below the left costal margin. A camera port is placed two fingerbreadths superior and left of the umbilicus. This can be adjusted for patients with very long torsos. Next, the right subcostal retraction arm port is placed in the same transverse plane as far laterally as is safe. An additional working arm port is placed splitting the difference between the first two. The camera is then placed in one of the right-sided ports to allow clear visualization of the left side. A left subcostal port is placed as far laterally as is safe, again in the same transverse plane as the prior three ports. An 11-mm assistant port is then placed, again splitting the difference between the camera and left subcostal port. Finally, a Nathanson retractor is placed just below the xyphoid process.
The patient is placed in slight reverse Trendelenburg and the robot is then brought into place for docking. Once all ports are docked, the robot arms are separated such that arms 1 and 2 are swung out to the patient’s right, and arms 3 and 4 are swung out to the patient’s left. This is critically important to allow enough space for the surgeon at the neck to proceed with the mediastinal portion of the procedure. The instruments are inserted under direct vision (Table 1).
Table 1
| Port | Size (mm) | Location | Hand | Instrument |
|---|---|---|---|---|
| Arm 1 | 8 | Right subcostal | Left | Small grasping retractor |
| Arm 2 | 8 | Right side between arms 1 and 3 | Left | Cadiere |
| Arm 3 | 8 | Two fingerbreadths superior and left of umbilicus | – | Camera |
| Arm 4 | 8 | Left subcostal | Right | Vessel sealer or long bipolar forceps |
| Assistant port | 11 | Left side between arms 3 and 4 | Assistant | Suction or laparoscopic grasper |
| – (no port is used for the Nathanson retractor) | – | Xiphoid | – | Liver retractor |
Technique
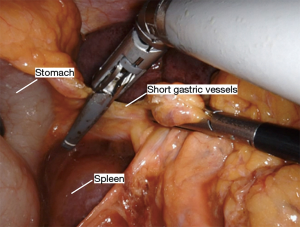
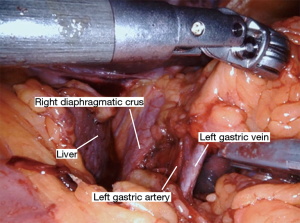
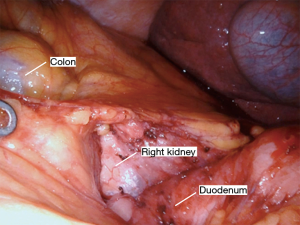
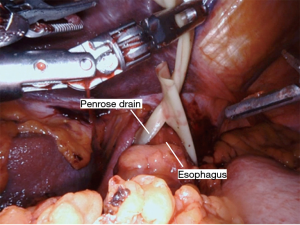
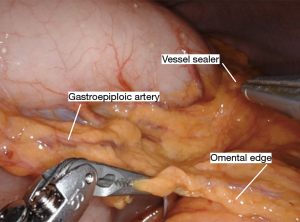
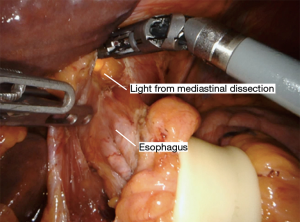
The Nathanson retractor is already positioned to elevate the liver from the hiatus. The small grasping retractor is used to elevate the stomach (Figure 1). The vessel sealer is then used to divide the omentum and enter the lesser sac away from the gastroepiploic artery (Figure 2). This dissection is continued up to the hiatus through the short gastric vessels (Figure 3). The stomach is now elevated further anteriorly and any retrogastric adhesions are divided. The left gastric vascular pedicle is identified for later division (Figure 4). Next, an aggressive Kocher maneuver is performed to ensure excellent mobility and reach of the conduit (Figure 5). We generally use the long bipolar forceps in the right hand for this purpose. Upon completion, the pylorus should reach the hiatus without any tension. The gastrohepatic ligament is then opened and dissection is carried through the hiatus into the mediastinum. A Penrose drain is passed around the gastroesophageal junction to allow the bedside assistant to help facilitate this dissection (Figures 6,7). This should connect circumferentially with the dissection performed from the neck. The left gastric artery should be divided at this point. This can also be accomplished before proceeding into the mediastinum. It can be divided either with the vessel sealer or a vascular load on a handheld laparoscopic stapler by the bedside assistant. At this point, the stomach should be completely mobile and this can be tested by observing the stomach while placing traction on the esophagus from the neck.
Video-assisted cervical portion
Cervical dissection
A low cervical incision of surgeon preference is created. This can be transverse, one fingerbreadth above the sternal notch, or oblique along the anterior border of the sternocleidomastoid. The platysma is divided, and Lonestar hooks are placed for exposure. These are used specifically because of their low profile, mobility, and versatility. These are adjusted as needed during the exposure of the esophagus. The remainder of the dissection is typically performed bluntly to avoid injury to nerves, and especially the left recurrent laryngeal nerve. Division of the omohyoid muscle is not necessary. The strap muscles are located and followed posteriorly to the cervical spine. The esophagus is circumferentially freed with digital dissection and encircled with a Penrose drain.
Mediastinal dissection
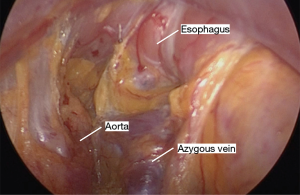
Specific instruments allow the mediastinal dissection to be performed under video assistance (Table 2). The plane posterior to the esophagus is developed first. This prevents dependent collection of blood and fluid if this step was performed later. The Penrose drain is retracted to the patient’s right shoulder, slightly elevating the esophagus anteriorly. The vein scope is introduced posterior to the esophagus, with its longitudinal fibers visible at the top of the screen anteriorly. The LigaSure is then used to dissect the esophagus away from the posterior mediastinum. Great care is taken to cauterize all bridging vessels, as even slight bleeding can be a nuisance. The aorta and azygous vein are typically well-visualized (Figure 8). The thoracic duct can sometimes be seen, but this is not intentionally identified. Dissection is carried as far distally as possible, preferably to the level of the hiatus. At this point, instruments are withdrawn.
Table 2
| Sorin VascuClear Endoscopic Vein Harvesting Kit (LivaNova, London, UK) |
| 5 mm, 30-degree thoracoscope |
| CO2 insufflation |
| LigaSure Maryland Jaw Laparoscopic Sealer (Medtronic, Minneapolis, MN) |
| Lonestar Retractor hooks (Cooper Surgical, Trumbull, CT) |
| Standard open-tip Yankauer suction, with suction pressure very low or controlled by an instrument |
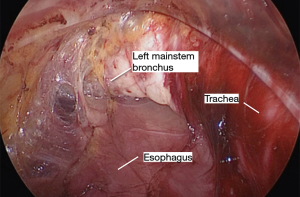
Next, the plane anterior to the esophagus is developed. The Penrose is retracted toward the left shoulder. The vein scope is introduced anterior to the esophagus, with its longitudinal fibers now at the bottom of the screen. The LigaSure is similarly used to carefully separate the esophagus from the anterior structures, including the airway and pericardium (Figure 9). This is again carried as far distally as possible. During this portion of the dissection, the scope can compress the airway and communication with the anesthesia team is important.
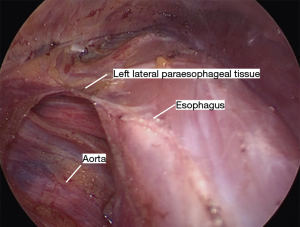
Finally, the anterior and posterior planes are connected by dividing the remaining bridging connective tissue on either side of the esophagus. This is typically best done starting proximal on the esophagus where the two planes are already joined, and continuing them distally (Figure 10).
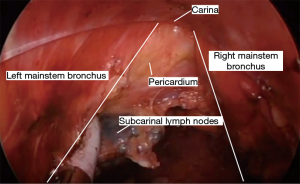
Once the esophagus has been fully mobilized, a lymph node dissection is performed. Any visible station 8 lymph nodes that were not mobilized with the esophagus are removed. Occasionally station 3 lymph nodes can be identified and easily removed. A station 7 dissection with this approach is challenging, but can usually be thoroughly accomplished with careful attention to hemostasis. The carina can be located by identification of the apex of station 7 lymph node tissue. The dissection is started in the apex and carried distally along both mainstem bronchi anteriorly to the pericardial surface. The lymph nodes in station 7 are then removed as a packet (Figure 11).
Anastomosis
The esophagus is divided at the neck, incorporating a very long 1-inch Penrose drain and ensuring 6 cm of esophagus can be brought out of the neck without tension for later anastomosis. The robot is undocked. A small upper midline laparotomy is created and a wound protector inserted. The specimen is delivered through the laparotomy. A gastric emptying procedure of choice may be completed at this point if desired. The conduit is then fashioned beginning on the lesser curve distal to the left gastric artery to include its lymph node supply. A series of heavy staple loads is used to create a 4-cm conduit parallel to the greater gastric curve, stretching the stomach appropriately with each staple load and finally separating the specimen from the conduit. The conduit is then transferred to the neck through the mediastinum in orthotopic position.
We prefer a 2-layer linear stapled anastomosis using a 60-mm staple load and oversewing the staple line. To accomplish this, the ends of the esophagus and gastric conduit are carefully retracted out of the neck adjacent to each other. A small gastrotomy is created and three stay sutures are placed approximating the two openings. The stapler is then inserted with the anvil on the gastric side and cartridge on the side of the esophagus. When the proper alignment has been achieved, the stapler is closed. A series of interrupted silk sutures is then placed along both sides of the stapler from the tip of the stapler to the orifice of the stomach and esophagus, thereby creating an outer handsewn layer. The stapler is then fired, creating the common channel. A nasogastric tube is then passed distally prior to closing the common channel with a Proximate TX stapler (Ethicon, Cincinnati, OH, USA) using a 30-mm green load and oversewing this.
The robot is again docked. The midline laparotomy is closed by clamping the wound protector shut between wet laparotomy sponges to allow for reintroduction of CO2 insufflation. The conduit’s course through the hiatus is inspected to ensure it appears straight and unkinked. A nonabsorbable polyfilament suture is then used to secure the conduit to both crura. This is performed to prevent both excessive conduit herniation through the hiatus, as well as bowel.
If a feeding tube is desired, the ligament of Trietz is identified and an ideal position for the jejunostomy tube is located for open jejunostomy insertion via the midline laparotomy. We do not routinely place feeding tubes in all esophagectomies as the complication rates from this procedure are low and the postoperative course is generally unremarkable.
Postoperative care
Patients are admitted to the stepdown floor postoperatively. They are maintained on IV fluids, and daily chest radiograph is obtained. The feeding tube, if in place, is used for medications. The nasogastric tube is placed to suction for 24 hours and is clamped on postoperative day 1 if drainage is low. If the conduit is not dilated and the patient experiences no nausea, the tube is removed on postoperative day 2. After evaluation by a certified speech language pathologist, a clear liquid diet is initiated on postoperative day 2 after nasogastric tube removal, and advanced to a soft diet as tolerated. All patients receive a speech therapy evaluation prior to initiating a diet due to higher risk of recurrent laryngeal nerve injury with a neck anastomosis. A contrast esophagram is not routinely obtained. Tube feedings are initiated on postoperative day 2 and advanced as clinically appropriate if a feeding tube was placed. Patients are typically discharged on postoperative day 5.
Conclusions
We describe a technique in performing a minimally invasive transhiatal esophagectomy, with a robot-assisted abdominal and video-assisted cervical approach. The main advantage of the robot-assisted abdominal approach is the magnified, stereotactic view provided by the camera and the increased dexterity provided by the instruments. Using a camera in both the abdominal and cervical portions of the procedure allows a complete thoracic lymph node dissection.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Christopher R. Morse and Uma M. Sachdeva) for the series “Minimally Invasive Esophagectomy” published in Annals of Esophagus. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aoe.2019.12.02). The series “Minimally Invasive Esophagectomy” was commissioned by the editorial office without any funding or sponsorship. DSD reports personal fees (Robotic Thoracic Surgery Proctor) from Intuitive Surgical, Inc., outside the submitted work. WBT reports personal fees (Robotic Thoracic Surgery Instructor and Observation Site Administrator) from Intuitive Surgical, Inc., outside the submitted work. NSL reports other (travel and courses) from Intuitive Surgical, outside the submitted work. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Orringer MB, Sloan H. Esophagectomy without thoracotomy. J Thorac Cardiovasc Surg 1978;76:643-54. [Crossref] [PubMed]
- Orringer MB. Transhiatal Esophagectomy: How I Teach It. Ann Thorac Surg 2016;102:1432-7. [Crossref] [PubMed]
- Orringer MB, Marshall B, Chang AC, et al. Two thousand transhiatal esophagectomies: changing trends, lessons learned. Ann Surg 2007;246:363-72; discussion 372-4. [Crossref] [PubMed]
- Horgan S, Berger RA, Elli EF, et al. Robotic-assisted minimally invasive transhiatal esophagectomy. Am Surg 2003;69:624-6. [PubMed]
- Dunn DH, Johnson EM, Anderson CA, et al. Operative and survival outcomes in a series of 100 consecutive cases of robot-assisted transhiatal esophagectomies. Dis Esophagus 2017;30:1-7. [Crossref] [PubMed]
- Galvani CA, Gorodner MV, Moser F, et al. Robotically assisted laparoscopic transhiatal esophagectomy. Surg Endosc 2008;22:188-95. [Crossref] [PubMed]
- Coker AM, Barajas-Gamboa JS, Cheverie J, et al. Outcomes of robotic-assisted transhiatal esophagectomy for esophageal cancer after neoadjuvant chemoradiation. J Laparoendosc Adv Surg Tech A 2014;24:89-94. [Crossref] [PubMed]
- Tangoku A, Yoshino S, Abe T, et al. Mediastinoscope-assisted transhiatal esophagectomy for esophageal cancer. Surg Endosc 2004;18:383-9. [Crossref] [PubMed]
- Mori K, Aikou S, Yagi K, et al. Technical details of video-assisted transcervical mediastinal dissection for esophageal cancer and its perioperative outcome. Ann Gastroenterol Surg 2017;1:232-7. [Crossref] [PubMed]
- Fujiwara H, Shiozaki A, Konishi H, et al. Transmediastinal approach for esophageal cancer: A new trend toward radical surgery. Asian J Endosc Surg 2019;12:30-6. [Crossref] [PubMed]
- Seto Y, Mori K, Aikou S. Robotic surgery for esophageal cancer: Merits and demerits. Ann Gastroenterol Surg 2017;1:193-8. [Crossref] [PubMed]
- Egberts JH, Schlemminger M, Hauser C, et al. Robot-assisted cervical esophagectomy (RACE procedure) using a single port combined with a transhiatal approach in a rendezvous technique: a case series. Langenbecks Arch Surg 2019;404:353-8. [Crossref] [PubMed]
- Nakauchi M, Uyama I, Suda K, et al. Robot-assisted mediastinoscopic esophagectomy for esophageal cancer: the first clinical series. Esophagus 2019;16:85-92. [Crossref] [PubMed]
Cite this article as: Demos DS, Tisol WB, Lui NS. Minimally invasive transhiatal esophagectomy. Ann Esophagus 2019;2:21.